Heisenberg principle of uncertainty12/6/2023 
In the published 1927 paper, Heisenberg originally concluded that the uncertainty principle was Δ pΔ q ≈ h using the full Planck constant. Introduced first in 1927 by German physicist Werner Heisenberg, the uncertainty principle states that the more precisely the position of some particle is determined, the less precisely its momentum can be predicted from initial conditions, and vice versa. Formulated by the German physicist and Nobel laureate Werner Heisenberg in 1927, the uncertainty principle states that we cannot know both the position and speed of a particle, such as a photon or electron, with perfect accuracy the more we nail down the particle's position, the less we know about its speed and vice versa. Specifically, it says that the more accurately you measure the. The Heisenberg uncertainty principle dates back to Heisenbergs work in Copenhagen in 1927, and although radical when it first surfaced, it is now well entrenched in literature and research based. Such paired-variables are known as complementary variables or canonically conjugate variables. The Heisenberg uncertainty principle is a law in quantum mechanics that limits how accurately you can measure two related variables. More formally, the uncertainty principle is any of a variety of mathematical inequalities asserting a fundamental limit to the product of the accuracy of certain related pairs of measurements on a quantum system, such as position, x, and momentum, p. The Uncertainty principle is also called the Heisenberg uncertainty principle. In other words, the more accurately one property is measured, the less accurately the other property can be known. And, there exists a minimum value for the product of the uncertainties of these two measurements. This principle says that the position and momentum of any particle cannot be simultaneously measured with arbitrarily high accuracy. 
It states that there is a limit to the precision with which certain pairs of physical properties, such as position and momentum, can be simultaneously known. This principle was given in 1927 by the German physicist Werner Heisenberg. The uncertainty principle, also known as Heisenberg's indeterminacy principle, is a fundamental concept in quantum mechanics. Uncertainty principle of Heisenberg, 1927. Trying to pin a thing down to one definite position will make its momentum less well pinned down, and vice-versa. 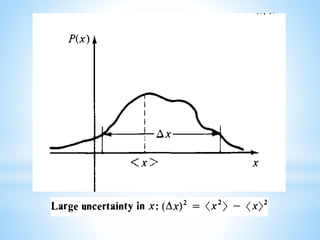
According to Heisenberg’s uncertainty principle, equation (1.27) x p > h / 2. Proposed in the 1920s by Werner Heisenberg, the. If any electron is confined within the nucleus then the uncertainty in its position (x) must not be greater than 10 -14 m. Werner Heisenberg stumbled on a secret of the universe: Nothing has a definite position, a definite trajectory, or a definite momentum. Theoretically, this should not be possible under the Heisenberg uncertainty principle, one of the most well-known tenets of quantum mechanics. Canonical commutation rule for position q and momentum p variables of a particle, 1927. The Uncertainty principle is also called the Heisenberg uncertainty principle.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
